18 May 2021
Nexstim granted authorization for commercial distribution of NBS system in Australia
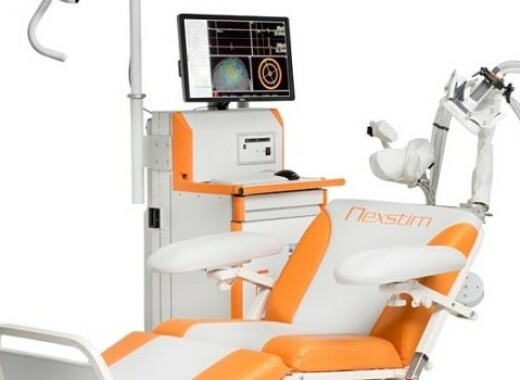
Nexstim Plc (NXTMH:HEX, NXTMS:STO) announces having received authorization for distribution of its NBS system by the Australian Therapeutic Goods Administration (TGA). The medical device license allows the company to start marketing and selling NBS systems in Australia to be used for neurosurgical applications. Nexstim’s NBS System is the only FDA cleared system based on navigated Transcranial Magnetic Stimulation (nTMS) for non-invasive mapping of the speech and motor cortices of the brain. The NBS system is used, for example, after a patient has had a diagnosis of a brain tumor or other disorder and when the lesion is thought to be close to functional areas of the brain - such as those responsible for speech production and limb movement. Brain maps from NBS can be invaluable when deciding the best treatment option for the patient.
Further information on Nexstim’s website.


